Ina Laura Perkins, CEO of Scandinavian Real Heart, talks about the groundbreaking technology behind the company's artificial heart, the results from the pre-clinical studies and how Real Heart plans to invest the funding from the current new share issue. Ina also shares the company's plans for the future.
Can you describe in detail the technology behind the artificial heart that Scandinavian Real Heart is developing? How does it differ from other artificial hearts currently on the market?
The totally artificial heart that we are developing - Realheart TAH - is based on Swedish research into what the human heart looks like and how it pumps. This means that our patented innovation is the world's first artificial heart that has two atria and two ventricles and therefore pumps like the human heart. The aim is to create a natural blood flow that is gentle on the blood and thereby reduce the risk of blood-related side effects that are common with artificial hearts. This is very important for doctors.
 Artificial heart pump Source: Scandinavian Real Heart
Artificial heart pump Source: Scandinavian Real Heart
What is important for the patient is to be able to live a normal life with an artificial heart, and therefore our solution is also silent, has a longer battery life and the accessories that the patient has to wear weigh less. But currently, treatment with artificial hearts means that the patient has an electrical cable that comes out through the skin, and as long as you have this cable, you cannot live a completely normal life. Our solution is energy efficient, which means that wireless charging, that is, getting rid of the cable, is a future possibility as a follow-up product.
Together with another Swedish company, we have started developing the first prototype of the wireless charging in a Vinnova-funded project. Another advantage of our solution is that we have designed our product for automated production to the greatest extent possible. With 64 million heart failure patients in the world, of which doctors estimate that there is an annual need for up to 60,000 hearts in Germany alone, it is important that we invest from the outset in making mass production a future possibility.
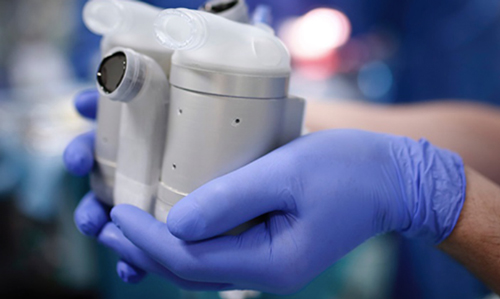 Artificial heart pump (Scandinavian Real Heart) Source: Lindköping University
Artificial heart pump (Scandinavian Real Heart) Source: Lindköping University
Can you tell us about the results of your preclinical studies? What are the most significant findings and how will they impact future development and testing?
For doctors to gain confidence in using the product, it is all about being able to demonstrate natural blood flow and good blood management. In lab tests where we connected the heart to a model of a patient, we have been able to show that Realheart TAH provides a pulsatile blood flow that can be fine-tuned with the same parameters as the human heart: pulse, stroke volume and percent systole. In lab tests with human blood, we have shown that the product has less than half the blood damage (hemolysis) compared to the market leading product. In animal studies, we have not seen any clinical blood damage (hemolysis) or blood clots. This is what doctors are primarily interested in.
In addition to the above, we have achieved 4 days of survival. Our French competitor achieved 4 to 10 days of survival before approval to start clinical trials. We are now working towards 10 days of survival and the cardiac surgeons who are carrying out our animal study say that some improvements in the surgical method are needed to reach our goal. Animal care is different from how you would take care of patients, for example you have to wake the animals up and they should stand up as quickly as possible, while a patient is kept under anesthesia and delicate new stitches are left to heal, so that is why we need to work a little on the surgical method to complete our animal study.
What advantages do you see with the Real Heart artificial heart compared to existing alternatives? What does this mean for patients?
A patient with severe heart failure needs a new heart. The first thing that comes to mind is a heart transplant. But there are not enough donated hearts. In Sweden, for example, 60 heart transplants are performed per year, while approximately 3,500 heart failure patients die. This is a great need that artificial hearts can help fill. An artificial heart can be on the shelf at the clinic and the doctors can insert it and save the patient's life, thereby buying time to plan care. You can either use the artificial heart while you are on the waiting list for a transplant, to ensure that you survive until you receive a donated heart, or you can continue with the artificial heart. One difference with a heart transplant is that you do not need immunosuppressive drugs, but only blood-thinning medication. Immunosuppressive drugs have many side effects and can lead to cancer. That is why a heart failure patient who is also a nurse told me that when she needs a new heart, she wants an artificial one.
Can you elaborate more on the company's view on biological heart transplantation? How is this view integrated into your products and research?
To reach patients, the first step is to offer the artificial heart to patients on the waiting list in a small clinical study. This means that for a limited time, a maximum of 6 months, data is collected from a smaller group of patients to show that the artificial heart is safe. After that, the next step is further clinical trials to make the heart available to patients who never even get the chance to be on the waiting list, as well as those in other countries. We are therefore now working towards the goal of being able to start a clinical trial that includes patients waiting for a transplant. This work is being carried out in collaboration with Sahlgrenska and means that we are collecting the preclinical data required to obtain regulatory approval to start a clinical trial.
What are Real Heart's primary objectives in using the funds from the current rights issue? How will these proceeds contribute to the company's long-term vision and strategy?
The main goal is to complete the preclinical studies in three areas:
- Lab tests with human blood
- Animal studies
- Mechanical endurance tests
 Scandinavian Real Hearts lab Photo: Impala Nordic
Scandinavian Real Hearts lab Photo: Impala Nordic
To do this, we need to involve another mechanical supplier and complete the clinical control unit (the computer that is on the outside and controls the heart). Once we have completed preclinical studies, it will open doors for other financiers who enter the later stage, for example, you can apply for loan financing from the European Investment Bank, they support innovative companies with up to 50 million EUR in commercialization loans. Our long-term vision is that artificial hearts, which are already a treatment that has been proven to save lives, will become as routine as inserting a pacemaker. This requires a good artificial heart that allows patients to live a normal life, and the ability to scale up production to make it available to everyone who needs it. This is what we will deliver.
Can you tell us more about your strategic partnerships? How will these collaborations support Scandinavian Real Heart's growth and innovation?
Developing an artificial heart requires expertise and experience, and to gather the best expertise, we have chosen to work with partners who have done similar projects before. That is why we are collaborating with Berlin Heart in Germany and with Hydrix in Australia.
Berlin Heart makes heart pumps that are on the outside of the body and are perhaps best known for being the only company to make heart pumps for children. Many children have been saved during the 30+ years that Berlin Heart has been supplying heart pumps to the global healthcare system. Berlin Heart makes the inside of the Realheart TAH itself in its factory in Germany. To ensure that the patient has the best experience and that the product is as safe as possible, we place great focus on the control unit and therefore we have chosen to collaborate with Hydrix in Australia who have experience from developing control units for 8 other heart pumps.
Finally. Where do you see Scandinavian Real Heart in 3 years?
Then we save the lives of heart failure patients and scale up our production to reach even more people.
Thanks to Scandinavian Real Heart and Ina Laura Perkins for the interview. We wish the company all the best in the future.
Disclaimer: This is an interview that has been done on behalf of the company. Impala Nordic or people behind Impala Nordic owns no shares in the company at the time of the interview.

