Pål Ryfors, CEO of Episurf Medical, looks towards 2023 and has his sights set on the US market and the commercialization of Episealer.
Who is Episurf Medical and can you briefly tell us about your business?
We are a medtech company that has developed a technology to treat bone and cartilage damage in the knee and ankle. Simply put, we treat patients with early signs of osteoarthritis, which is a large patient group that today largely lacks effective treatments.
What is the biggest difference between you and your competitors?
For the patient group we target, there is currently no so-called "gold standard", which means that we are compared to many different types of treatments, and in this comparison, our strong clinical data is our absolute strongest card.
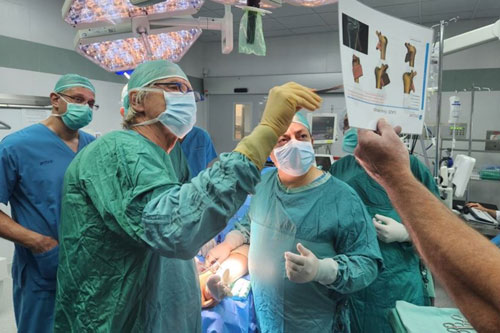
A couple of weeks ago, the first operation with Episealer was performed in Israel. Can you tell us a little more about the operation in detail and what the outcome means for you?
This particular operation was our first ankle surgery in Israel, we have previously performed knee surgery in the country. It was a very prominent orthopedic surgeon who, together with one of our clinical advisors, operated on a patient with major foot problems.
A very successful operation with great interest. It means a lot as the first operation in a new country is always the most difficult to achieve. But it means that you cross a threshold and that you have a new platform to work from.
At the end of September, you received feedback from the FDA that the approval process is proceeding as planned. Can you tell us a little more about the planned activities to be carried out in Q4 to resolve the remaining questions that the FDA has asked about?
It is an interactive work with the FDA where you carefully review the feedback you receive. Some things need to be clarified, i.e. it may be a matter of pure misunderstandings, some things need to be clarified in one way or another through, for example, additional scientific references, and some things may need to be changed. We try to maintain as close a dialogue with the FDA as possible, and together with skilled advisors and a committed team, we should be able to get the pieces in place. Hard work, simply put.

What does a final FDA approval mean in brief for Episurf Medical?
This would be our first approval to bring a product to the US market, which would be a major milestone. However, this is just one of several products that we have on our way to the US, and from that perspective, it is just the beginning. But it is undeniably an important milestone as we can begin commercializing in the US.
What does the market potential look like globally and how large a market share could a player like Episurf Medical take in the future?
The Joint Reconstruction market is estimated at around $18 billion globally, with knee making up the largest portion, around half ($9 billion). We are not targeting the entire patient group, but we are talking about a TAM (total addressable market) of around $1 billion globally, and we are now adding more products in both knee and ankle. The market leaders in orthopedics have a market share of around 30%, and we are not aiming lower than that. It is of course a long-term market share goal, as we have just started commercializing. But we believe that we have a huge opportunity commercially if we manage to capitalize on the strong data we already have.
Looking ahead to 2023, what would you say is your biggest focus?
There are two main priorities;
The first is the US market. Partly to get our first product on the market (Episealer Patellofemoral System), partly to move forward with our big toe solution (Episealer MTP-System) and to make progress in the US study for Episealer Knee (EPIC Knee trial).
The second part has to do with our commercialization outside the US. We have done operations in about 20 countries in the last 12 months and we have good customer growth right now. Now we just have to increase the pace further and make sure that we really make a commercial mark in the market. The platform is in place for this, so it's just delivery that matters.
What are your sales goals?
We do not provide any short-term goals or short-term guidance except that we have said that we are aiming for high growth in the markets where we have begun commercialization.
When can Swedish healthcare expect to use Episealer on patients?
It is already being done! However, on a fairly limited scale. But our first patient was operated on in Stockholm almost 10 years ago, and he is doing absolutely fine today. We are right on the border between experimental surgery and established practice, and we believe that in the future more and more Swedish doctors will open their eyes to us, to the benefit of more and more patients in Sweden.
We thank Episurf Medical and Pål Ryfors for the interview and wish the company great success in the future!
Impala Nordic, or the people behind Impala Nordic, do not own any shares in the company.

